
- 3876 Industrial Ave, Rolling Meadows, IL 60008
- Info@auctuselectro.com
- 3876 Industrial Ave, Rolling Meadows, IL 60008
- Info@auctuselectro.com

Medical Cable Assembly
- Home
- Medical Cable Assembly

Introduction
Every little thing matters in the medical field. One connection failure could obstruct diagnostic or therapeutic acts or even threaten a patient. Auctus Electro specializes in custom cable harnesses and medical cable assembly applications in the field of healthcare. Patient monitoring, diagnostic imaging, or surgical equipment-contained performance you can trust from our cables and assemblies.
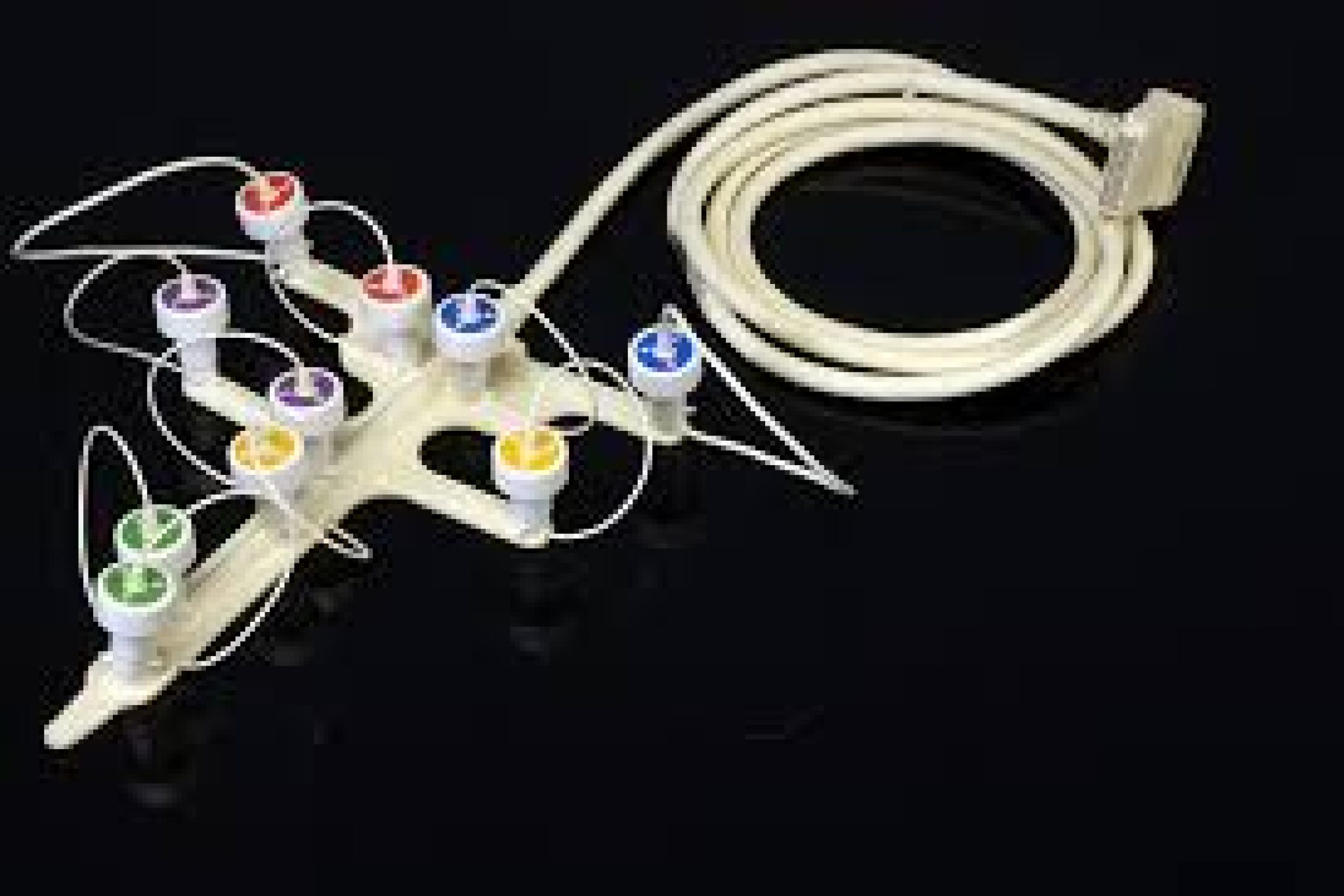

What is a medical cable assembly?
A medical cable assembly consists of a bundled assembly of wires or cables terminated with connectors and jacketed and protected to ensure safe use with medical equipment. While a general cable may carry power or data, a medical-grade assembly must satisfy additional requirements: durability, tolerance to sterilization, biocompatibility, shielding, and safety.
These assemblies are used to join imaging equipment, sensors, monitors, and surgical or patient lead devices.
Why choose specialised medical cable assemblies over standard cables?
Cable standardization may apply, but the requirement in the medical field is very strict. Examples of such were given:
Patient contact: Materials have to be safe, non-toxic, and free from any harmful reactions.
Sterilization: Several assemblies need to tolerate steam autoclaving, chemical disinfection, or radiation sterilization.
Signal Integrity: For imaging systems, monitoring devices, or diagnostics, the minimum inducing electromagnetic interference (EMI/RFI).
Flexibility & durability: Conducted flexions, movements, and repetitive cleaning demand that cables maintain performance over time.
Compliance: Standards such as ISO 13485, IEC 60601, and ISO 10993 play a crucial role in the manufacturing process.
Due to these factors, designing and manufacturing medical cable assemblies is more than just bundling wires: it’s an engineering challenge.
Applications & Use Cases
Cables for medical equipment appear in different aspects of health technologies. The most popular ones are
Patient Monitoring & Diagnostic Leads
Cables connect ECG/EEG, pulse oximeters, and other bedside equipment as part of the patient-interface assemblies. Biocompatibility and flexible construction are required in this case to ensure patient comfort and long-term durability.
nai-group.com
Ultrasound, MRI, CT, and endoscopy imaging systems
High-performance medical cable assemblies form parts of ultrasound probes, MRI or CT systems, or endoscopic devices. They may carry high-frequency signals, have fiber optics, have very low tolerances required by the cable assemblies, or need shielding to have a good image.
Surgical & Interventional Devices
They can be found in mini-procedural cables or surgical suites. In this, cable assemblies are interfacing sensor robotics and catheters with the rest of the devices. They also need sterilizability, miniaturization, flexibility, and terminations.
Devices & Sub-assemblies
Large medical equipment (like imaging scanners and lab automation systems) makes use of internal cable assemblies for power, control, and data. They may not always come in contact with patients, but they still need to fulfill high reliability standards and compliance.
nai-group.com
Key Features & Technical Considerations
The following high-level features should be placed under scrutiny while selecting or designing a medical cable assembly. Below are some key things on which we at Auctus Electro would focus in every project.
Material & Jacket Options
Biocompatibility: For direct or indirect patient contact, materials should comply with standards such as ISO 10993 for cytotoxicity, irritation, and sensitization.
Sterilization Resistance: Materials should withstand repeated sterilization cycles—steam, EtO, plasma, or radiation. Such examples of materials are PTFE, polyurethane, and silicone.
Chemical & Abrasion Resistance: The outer jacket should be tough against cleaning agents, disinfectants, and mechanical wear.
Flexibility & Kinking Resistance: Flexibility would be necessary while using handheld or moving applications. The cable should also avoid kinking and resist fatigue.
Conductors, Shielding & Signal Integrity
Shielding is done mainly by either a braided or oil shield in the assembly to block off EMI/RFI and protect sensitive medical signals.
Fine gauge and micro-coaxial wires: Very fine wires are employed in many assemblies requiring precision termination and insulation (e.g., for sensors or cameras).
Hybrid assemblies: In order to save space and simplify routing, some systems carry both power and signal in one cable bundle.
Connectors & Overmolding
Connector choice: Sealing or overmolding for protection from fluid, sterilization, or repeated flexing is considered to enhance the robustness and reliability of the termination.
Overmolding: The majority of the medical cable assemblies undergo medical-grade overmolding to strengthen the junction between cable and connector while preventing bacterial growth.
Testing Certification & Compliance
Quality standards: ISO 13485 (medical devices), IEC 60601-1 (safety of medical electrical equipment), and IPC/WHMA-A-620 (cable-assembly workmanship), to name a few.
Validation & testing: Mechanical, electrical, environmental, and lifecycle testing to ensure assemblies meet performance and safety demands.
Traceability & documentation: For regulated environments, documentation of materials, processes, and test results is important.
Customisation & Design Process
Auctus Electro custom medical cable assemblies are designed to realize that no two medical device applications are alike.
1. Requirement-gathering
This is where we start understanding your application: is it through patient contact or through equipment only? What are the expected length, flexibility, signal/power needs, and method of sterilization? Will this environment see the cable? All of these will help in indicating secondary keywords that are important, e.g., medical device wiring harness, sterilizable medical cable, biocompatible cable assembly, etc.
2. Design & Prototyping
Based on requirements, we propose conductor types, shielding, jacket materials, connector types, overmolding, or strain relief options. We build prototypes for form-fit, function, and clearance testing. We pay special attention to miniaturized designs (fine wire, micro-coax) and assemblies for constrained spaces.
3. Validation & Documentation
On prototype approval, we validate the products via flex tests, mechanical pull tests, sterilization simulations, EMI tests, and electrical continuity checks. All to be documented for regulatory purposes.
4. Manufacturing & Quality Assurance
Strict quality control protocol is maintained in our production lines. Inspection and testing go for each cable assembly. Traceability of all components is maintained, and the best practice in harness assembly, overmold paths, shielding terminations, and labeling is applied.
5. Supply Chain & After-market Support
We cater to low-volume, high-complexity builds and high-volume production runs. We provide aftermarket service (replacements, variants, or revised builds). In regulated medical environments, a supply partner with excellent after-market support is imperative.
Selecting the Right Medical Cable Assembly Supplier
When selecting a partner for medical cable assemblies, several questions should arise:
- ISO 13485 (or equivalent) certification for the manufacture of medical devices was in place?
- Can they supply materials validated for biocompatibility (ISO 10993) and sterilization?
- What experience do they have in healthcare applications (imaging, monitoring, surgical)?
- Do they support customization (i.e., fine wire, hybrid assemblies, micro-coax, over-molding)?
- How are traceability, documentation, and testing done?
- What are their lead times and stability of supply chain (especially for high-volume medical OEMs)?
- Do they support aftermarket work, revisions, and lot traceability?
Auctus Electro satisfies all the requirements: serving industrial and medical domains, being highly qualified to deliver custom medical cable assemblies whilst maintaining compliance and reliability.
Summary
The bottom line is, if your device or equipment requires reliability, sterilization compatibility, patient-safe materials, and high signal/power integrity, you need medical cables and cable assemblies, not commercial cables. By Auctus Electro, you can rely on the expertise, manufacturing capability, and quality mindset to make these assemblies—from prototype to production—along with all necessary documentation and support. Whether it’s the monitoring systems or imaging equipment, surgical instruments, or diagnostic devices, we cater to all your cable assembly needs.

